Beyond Brain-Eating Amoebas: Current Insights into Naegleria fowleri Identification, Prevalence, and Treatment
© 2025 Bio Communications
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Abstract
Naegleria fowleri, commonly known as the “brain-eating amoeba,” is an opportunistic, free-living protozoan responsible for primary amoebic meningoencephalitis (PAM), a rare but almost invariably fatal infection of the central nervous system. This thermophilic amoeba thrives in warm freshwater environments such as lakes, hot springs, and poorly maintained swimming pools. It can survive under high temperatures, making it more prevalent in regions with warm climates and developed countries, including Australia, Europe, and the United States. This review summarizes current evidence on the global prevalence, clinical presentation, diagnostic challenges, and treatment strategies related to N. fowleri infections. Epidemiological data indicate that approximately 26.4% of global freshwater sources contain Naegleria species, with the highest prevalence recorded in the Americas at 33.2%. Infected individuals typically present with acute symptoms such as severe headache, high fever (101–105°F), neck stiffness, seizures, altered mental status, and coma, with disease progression often resulting in death within one to two weeks of symptom onset. Despite advances in supportive care and antimicrobial therapy, the case fatality rate remains over 90%. Conventional diagnostic methods include microscopy, culture, serology, imaging, and molecular techniques such as real-time PCR; however, delays in diagnosis often hamper timely treatment. Therapeutic regimens traditionally involve a combination of antifungal and antimicrobial agents, including amphotericin B, azoles, azithromycin, and miltefosine. Although amphotericin B is considered the gold standard due to its potent amoebicidal activity at concentrations as low as 0.01 μg/ml, its poor solubility and high toxicity can cause significant adverse effects such as nephrotoxicity, hematologic complications, and systemic symptoms like headache, vomiting, and fever. Additionally, in vitro studies have shown that N. fowleri may develop resistance to miltefosine at concentrations up to 40 μg/ml. This review underscores the critical need for early and accurate diagnostic tools, safer and more effective therapeutic alternatives, and greater public awareness to prevent exposure and improve patient survival. Further research is urgently required to develop novel treatment options and reduce the devastating impact of this neglected but deadly pathogen.
Keywords:
Meningoencephalitis, Molecular Diagnosis, Naegleria fowleri, Miltefosine, Amphotericin B.
1. Introduction
Naegleria fowleri, commonly referred to as the “brain-eating amoeba,” is a thermophilic, free-living protozoan that causes primary amoebic meningoencephalitis (PAM), a rare but almost universally fatal infection of the central nervous system (CNS) (Datta, 2024; Kurup et al., 2022; Sriram & Chandra, 2024) . This amoeba thrives in warm freshwater environments such as lakes, hot springs, rivers, inadequately chlorinated swimming pools, and poorly maintained water distribution systems (Shirsath et al., 2024). Although often perceived as geographically limited, N. fowleri has been reported worldwide, including in the Americas, Europe, Asia, Africa, and Australia (Alanazi et al., 2025). A systematic review by Gharpure et al. (2020) documented cases in diverse climates, demonstrating that rising global temperatures may expand its geographic range, posing new risks in previously unaffected areas. Despite its environmental ubiquity, human infections remain rare, with fewer than 500 reported cases globally (Capewell et al., 2015). However, the high fatality rate, rapid disease progression, and lack of effective therapeutic options have made PAM a subject of growing concern (Mungroo et al., 2019). Most infections occur during recreational water activities in warm months when water temperatures exceed 25°C (Stahl & Olson, 2020).
While numerous studies have described the organism’s biology and global prevalence, inconsistencies in surveillance methods and underreporting hinder accurate estimation of its true distribution. There is also insufficient comparative analysis of diagnostic and therapeutic practices across regions, which limits our ability to identify critical gaps in prevention and treatment. This review aims to synthesize current knowledge of N. fowleri biology, prevalence, clinical features, diagnostic strategies, and therapeutic challenges, highlighting unresolved questions and emerging directions for future research.
2. Diagnosis of Primary Amoebic Meningoencephalitis
Early and accurate diagnosis of Naegleria fowleri infection is critical due to the rapid progression and high fatality of PAM (Phung et al., 2025). However, diagnosis remains challenging, as initial clinical symptoms often mimic bacterial or viral meningitis, leading to misdiagnosis or delayed treatment (Siddiqui et al., 2025). Several diagnostic methods have been developed, each with distinct advantages and limitations.
2.1. RT-PCR
RT-PCR for the recognition of N. fowleri proved its sensitivity and specificity; thus, it is a useful substitute for cultivation and microscopy (Saberi et al., 2020). But there are some disadvantages; such as being expensive (RFLP), nonetheless, it overcomes the late and retroactive diagnosis (ELISA), time-consuming (cultivation), or incomplete results (flagellation test) for the diagnosis of both environmental and analytic samples (Madarova et al., 2010). One of the fundamental benefits is the probability of separating DNA from specimens without preceding culture (Maclean et al., 2004). Also, PCR helps the identification of remaining spp. of the Naegleria genus (Madarova et al., 2010). Such particular tests for N. fowleri should be endorsed to verify suspected contagious mediums in analytic samples (Madarova et al., 2010). Nevertheless, their use is often limited by availability in resource-limited settings and the need for specialized equipment and trained personnel.
2.2. Imaging Methods
Magnetic Resonance Imaging (MRI) shows abrasions, or Computed Tomography (CT) scans the abrasions, but these are general overviews (Kidney & Kim, 1998; Schumacher et al., 1995; Singh et al., 2006). On the other hand, CT scans reveal the eradication of the cisterns encircling the subarachnoid space and the midbrain (Martinez, 2019). MRI or CT scans can help identify brain edema and inflammation but do not specifically differentiate PAM from other forms of meningoencephalitis (Mahajan & Mahajan, 2024). Thus, imaging primarily supports differential diagnosis and treatment planning rather than confirming N. fowleri infection.
2.3. Serology
Antibodies of Naegleria spp. had been detected in serologically active people (Marciano-Cabral & Cabral, 2007; Marciano-Cabral et al., 1987). Serological tests are not always fruitful because PAM is a swift disease. Usually, no increase is recorded in antibody titers, except within victims that were effectively examined and recovered from the inflammation (Vargas-Zepeda et al., 2005). Moreover, serological tests are generally of limited clinical value for PAM diagnosis, as they often fail to detect infection in the acute phase when rapid treatment decisions are needed (Christopher-Hennings et al., 2019).
2.4. Culture Methods
Biopsied brain tissues or cerebrospinal fluid (CSF) should be transferred to the diagnostic laboratory and kept at room temperature. This material can be implanted onto the micropropagation cells and cultivated at 37 °C in the existence of the antiseptics, e.g., streptomycin and penicillin (Mungroo et al., 2020). The available amoeba will replicate and damage the monolayers in 24-48 hours (Schuster, 2002). Autopsy tissue can also be spotted on 1.5% non-nutrient agar covered with a film of bacteria (E. coli) (Martinez, 1985; Martínez et al., 1980; Martinez & Visvesvara, 1991; Schuster, 2002). The amoebae will develop from the tissue, absorb the bacteria, and replicate (Hundt & Ruffolo, 2005). Using an inverted light microscope, amoeba can be observed on agar in the form of flagellates (Martinez, 1985) as shown in Fig. 1.
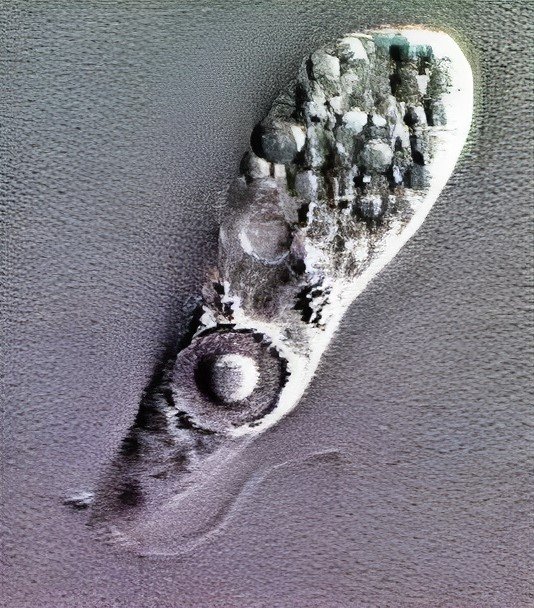
These techniques typically using non-nutrient agar with an Escherichia coli layer, can confirm viability and support species identification (Değerli et al., 2020). However, culture is time-consuming and may not provide timely results for urgent clinical management.
2.5. Microscopic Methods
An antemortem examination is very uncommon, but when cerebrospinal fluid pressure is low, a spinal tap can be achieved (Martinez & Visvesvara, 1991). The cerebrospinal fluid is slightly hemorrhagic and cloudy, with elevated cellularity and consisting mostly of neutrophils (Di Terlizzi & Platt, 2009). CSF is described by elevated protein and low glucose (Seehusen et al., 2003). The direct microscopic examination of cerebrospinal fluid as a wet mount is the technique of choice for PAM examination (Martinez & Visvesvara, 1991). CSF consists of moveable amoebae, which can be identified by light microscopic examination (Martínez et al., 1980; Martinez & Visvesvara, 1991, 1997). When amoebae are implanted in water, they can transform into swimming flagellates (Fig. 1); N. fowleri was distinguished from other brain and spinal cord infections causing free-living amoeba (FLA). If available in cerebrospinal fluid, amoebae can be recognized by staining fixed preparations with Wright's or Giemsa (Fig. 2). However, gram stain is utilized in scientific labs for the identification of bacteria in cerebrospinal fluid, but gram stain is not functional for the examination of amoebae (Martinez, 1985; Martínez et al., 1980; Martinez & Visvesvara, 1991; Trabelsi et al., 2012).
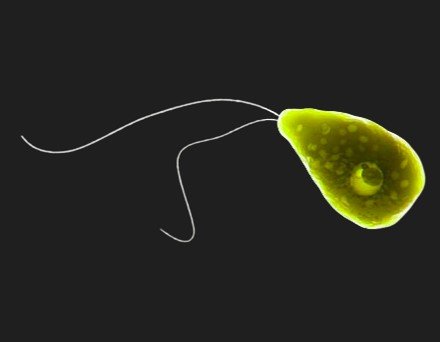
Although microscopy is quick and inexpensive, its accuracy heavily depends on sample handling, the examiner’s expertise, and parasite concentration in the CSF.
3. Prevalence and Epidemiology of N. fowleri
The global distribution of Naegleria fowleri is more widespread than commonly recognized (Gabriel et al., 2019; Maciver et al., 2020; Reyes-Batlle et al., 2019; Saberi et al., 2020; Siddiqui & Khan, 2014; Trabelsi et al., 2012), yet the true burden of human infections remains underestimated due to underreporting, limited surveillance, and misdiagnosis (Alanazi et al., 2025). A meta-analysis by Saberi et al. (2020) found that Naegleria species are present in approximately 26–30% of freshwater bodies worldwide, with the highest detection rates reported in North and South America (up to 33%) and increasing reports from Asia and Africa.
Despite this environmental prevalence, PAM remains a rare disease, with fewer than 10 reported cases annually in the United States and sporadic cases elsewhere (Diaz, 2012). This apparent inconsistency can be partly explained by the pathogen’s specific transmission route: infection occurs when contaminated water forcefully enters the nasal passages during activities such as swimming or diving, allowing the amoeba to migrate along the olfactory nerves to the brain (Capewell et al., 2015). Routine ingestion of contaminated water does not cause infection.
Climatic changes and urbanization are likely to expand the geographical range of N. fowleri by warming freshwater habitats and affecting water treatment infrastructure (Stahl et al., 2025). Notably, isolated reports of N. fowleri in non-traditional habitats, including poorly chlorinated tap water and geothermal power plant runoff, suggest the need for robust environmental monitoring and maintenance of water quality standards (Bartrand et al., 2014).
Current surveillance data remain fragmented, with most studies relying on environmental sampling without standardized protocols. Additionally, reliable data from developing countries is scarce due to limited diagnostic capacity and reporting systems. Therefore, improving systematic surveillance, especially in underreported regions, is essential to accurately map the risk of exposure and guide public health interventions.
This synthesis highlights the need for harmonized monitoring strategies, incorporation of molecular detection in routine water testing, and heightened clinical awareness in both endemic and emerging regions. Table 1 shows a comprehensive review of the previous studies conducted on the Prevalence of Naegleria spp. in origins of water across various countries.
| Country | Prevalence | Year | Methods | Sampling Year | Sample Number | References |
|---|---|---|---|---|---|---|
| Taiwan | 14.5% | 2010 | PCR | 2007–2009 | 62 | (Huang & Hsu, 2010) |
| USA | 26.67% | 2010 | PCR | 2006–2008 | 30 | (Laseke et al., 2010) |
| Italy | 72.00% | 2017 | PCR | 2013–2015 | 25 | (Di Filippo et al., 2017) |
| South Africa | 1.00% | 2017 | PCR | 2016 | 100 | (Muchesa et al., 2017) |
| Spain | 10.00% | 2017 | PCR | 2016 | 10 | (Reyes-Batlle et al., 2017) |
| Pakistan | 25.00% | 2017 | PCR | 2016–2017 | 120 | (Tanveer et al., 2017) |
| Egypt | 26.00% | 2018 | PCR | 2016–2017 | 102 | (Abd El Wahab et al., 2018) |
| Philippines | 26.00% | 2019 | PCR | 2017–2018 | 54 | (Milanez et al., 2019) |
| Egypt | 23.79% | 2023 | PCR | 2021–2022 | 165 | (Elseadawy et al., 2023) |
| France | 16.51% | 2011 | Nested PCR | 2009–2010 | 103 | (Ahmad et al., 2011) |
| Egypt | 38.02% | 2014 | Morphology | 2012–2013 | 250 | (AI-Herrawy et al., 2014) |
| Egypt | 0.89% | 2014 | Morphology | 2012 | 112 | (Khalifa et al., 2014) |
| USA | 30.14% | 2003 | ELISA | 2000–2001 | 71 | (Reveiller et al., 2003) |
| USA | 66.67% | 2013 | qPCR | 2011–2012 | 9 | (Painter et al., 2013) |
| Turkey | 2.7% | 2021 | qPCR | 2018–2020 | 111 | (Aykur & Dagci, 2021) |
| Korean | 11.54% | 2018 | Real-time PCR | 2016–2017 | 52 | (Kim et al., 2018) |
| Thailand | 1.47% | 2004 | Zymogram patterns | 2003 | 68 | (Tiewcharoen et al., 2004) |
| Malaysia | 29.00% | 2019 | Plating assays/PCR | 2017–2018 | 45 | (Gabriel et al., 2019) |
| Mexico | 7.00% | 2014 | Morphology/Plating assays | 2013 | 43 | (Bonilla-Lemus et al., 2014) |
| Spain | 14.28% | 2019 | Morphology/PCR | 2018 | 21 | (Reyes-Batlle et al., 2019) |
| Iran | 54.00% | 2017 | Morphology/PCR | 2015 | 50 | (Latifi et al., 2017) |
| Thailand | 9.00% | 2018 | Morphology/PCR | 2016–2017 | 33 | (Tiewcharoen et al., 2018) |
| Sri Lanka | 23.07% | 2020 | Plating assays/PCR | 2019 | 78 | (Gunarathna et al., 2021) |
| India | 20.7% | 2022 | Microscopy/NNA/PCR | 2020–2021 | 29 | (Krishnamoorthi et al., 2022) |
4. Prevalence of PAM in Pakistan
The accessible data revealed that the number of died people due to PAM has enhanced in few previous years. Despite that PAM first record was in 2008, till 2018, 13 deaths were recorded in Pakistan. Fifteen deaths were recorded in 2019 until October (Tabassum et al., 2022). There were no deaths recorded in 2020. Six deaths were recorded in 2021. However, first death due to PAM for the year 2022 was recorded on 2nd of May. The victim was from Kemari (Karachi) who is 59-year-old. Four cases of PAM were recorded until July 1, 2022. By the reports of to Dr. Bahoto, Director-General of the Sindh Health Department, all 4 victims had no records of entertaining water practices, and they were probably affected by household water supply (Tabassum et al., 2022). People from 26-45 years old were mostly affected by PAM in Pakistan (Naqvi et al., 2016).
In the summer, PAM cases are generally reported from April to September (Naqvi et al., 2016). Recently, it was revealed that samples of water were taken from several zones all over Karachi, along with two major sources and the zones provided by these distribution systems. Specimens of water were sent to identify N. fowleri via culture and PCR, which came back positive (Ghanchi, 2018). This is astonishing, given that the water in Karachi is frequently saline, and amoebas are not able to develop in saline water, revealing that the N. fowleri strain experienced in Pakistan is definite from those recorded worldwide, or it may have developed a defiance to the briny ecosystem (Ali et al., 2020) .
Moreover, a recent survey analysis by Aurongzeb et al. (2025) reported that PCR analysis using ITS- and Naegl-primers showed that N. fowleri was present in 11 out of 39 environmental samples (tap water) from Karachi. The main areas where N. fowleri identification was done included Lyari and Golimar areas. So, Aurongzeb et al. (2025) suggested that the presence of coliforms, high temperatures, and inadequate chlorination may all contribute to the presence of N. fowleri in these areas of Pakistan.
5.1 Treatment of PAM due Naegleria fowleri
Recently, The most utilized drug for the treatment of N. fowleri infections is Amphotericin B (AmB), an antifungal agent that kills amoebas by activating an apoptosis-like programmed cell death (PCD) (Cardenas-Zuniga et al., 2017). Formerly, the intrathecal or interveinal regime of AmB alone or conjugated with other medicines including Azithromycin (AZM), Rifampin (RIF), and Fluconazole (FCZ) has revealed some effectiveness in eradicating the amoeba when given in primary phase of the disease (Bellini et al., 2018; Grace et al., 2015). Furthermore, Miltefosine (MLT), an alkylphosphocholine compound utilized initially to control leishmania infections and cancer of chest in women (Verweij et al., 1992). It was thought to be a rare therapeutic drug used for the treatment of PAM until it was effectively treated; a 12-year-old child who withstand the infection with a complete neurological rehabilitation (Heggie & Kupper, 2017; Rice et al., 2020). This treatment protocol was later utilized on two distinct victims of PAM, where one of them got through with a poor neurological condition and the other could not survive (Cope et al., 2016). Though MLT is an assuring drug used for the cure of PAM, it does not guarantee complete rehabilitation from the infection (Cope et al., 2016).
5.2. Amphotericin B
There have been zero clinical trials to date that examine the potency of one treatment administration over another due to the rarity of N. fowleri infections in humans (Kim, Lee, et al., 2008). Most of the information concerning medication effectiveness was established through either in vitro studies or case reports (Kim, Lee, et al., 2008). The 0.01 μg/ml of amphotericin B is considered the minimum amoebicidal concentration utilized to control N. fowleri (Goswick & Brenner, 2003; Kim, Jung, et al., 2008). On the other hand, in vitro studies revealed that 0.1 μg/ml concentration of amphotericin B was required to decrease over 90% of N. fowleri development (Goswick & Brenner, 2003), while 0.39 μg/ml was required for the complete suppression of amoeba reproduction (Barnett et al., 1996). The suggested period of therapy is 10 days when amphotericin B is utilized for the medication of N. fowleri (Barnett et al., 1996; Hannisch & Hallagan, 1997). Amphotericin B (AmB) is not soluble in water (Torrado et al., 2008), therefore, it is very toxic and causes hematopoietic effects, electrolyte imbalances, renal toxicity, and other clinical symptoms like headache, nausea, chills, fever, and vomiting (Laniado-Laborin & Cabrales-Vargas, 2009). These complications clarify the importance of finding a safer and more effective alternative that can be used against N. fowleri (Cardenas-Zuniga et al., 2017; Debnath et al., 2012).
5.3. Miltefosine
Two victims of diseases caused by N. fowleri recovered after being cured with miltefosine in 2013 (Capewell et al., 2015). It is made up of phosphate and lipids (Dorlo et al., 2012) which are also attached to choline (Fig. 3). The detailed structures of miltefosine are amphiphile in nature, containing an aliphatic tail and a head region consisting of phosphate and choline (Grace et al., 2015). The mechanism of miltefosine action was proved as an inhibitory effect against protein kinase B (Dorlo et al., 2012).
Treatment of primary amoebic meningoencephalitis caused by N. fowleri is a challenging task because the disease is not only uncommon, but also has a low morbidity and high mortality rates (Visvesvara, 2013) . Some of the antifungal drugs clinically approved can be utilized to control infections due to Naegleria species, e.g., Fluconazole, Azithromycin, and Rifampin (Grace et al., 2015) . This is because the sterol biosynthesis pathway in infectious fungi is same in free-living amoeba (Debnath et al., 2017) .
6. Recent Therapeutic Approaches and Emerging Treatments
Recent research has focused on drug repositioning, nanoparticle delivery systems, and combination therapies to enhance efficacy and reduce toxicity (Siddiqui et al., 2024; Siddiqui et al., 2025). Several in-vitro and in-vivo studies have shown that repurposed FDA-approved drugs, such as statins, antipsychotics, and anticancer agents, may exhibit anti-amoebic activity, providing promising leads for combination regimens (Elsheikha et al., 2020).
Nanoparticle-based formulations of amphotericin B and other drugs have been explored to improve blood–brain barrier penetration, increase bioavailability, and reduce systemic toxicity (Siddiqui et al., 2024). For example, liposomal amphotericin B has demonstrated enhanced efficacy in experimental models, although human data remain limited (Adler-Moore et al., 2019). Other experimental approaches include silver and gold nanoparticles, which have shown amoebicidal effects in vitro (Anwar et al., 2019).
7. Conclusion
Primary amoebic meningoencephalitis caused by Naegleria fowleri remains one of the most challenging infectious diseases due to its rarity, rapid progression, and extremely high fatality rate despite aggressive treatment. Although the environmental presence of N. fowleri is well established across diverse freshwater sources globally, accurate assessment of human risk is still hampered by inconsistent surveillance, limited diagnostic awareness, and underreporting, especially in low- and middle-income countries. Current diagnostic methods, while advanced in laboratory settings, are not always practical or widely accessible for early clinical detection. Similarly, despite amphotericin B and miltefosine offering some therapeutic promise, their limitations in terms of toxicity, resistance, and delivery to the central nervous system continue to hinder treatment success. To meaningfully reduce the burden of PAM, several areas require urgent attention. First, robust and harmonized surveillance systems should be developed, particularly in regions with inadequate water treatment infrastructure and cultural practices that may increase nasal exposure. Second, clinical diagnostic protocols should integrate rapid, point-of-care molecular tools alongside improved training for healthcare professionals to recognize this rare disease early. Future therapeutic strategies must prioritize the development and clinical validation of new compounds and delivery systems, including nanoparticle-based formulations and drug repositioning approaches, to overcome the barriers posed by the blood–brain barrier and drug toxicity. Collaboration across microbiology, nanotechnology, and pharmaceutical research is essential to translate promising in vitro findings into viable treatments. Lastly, coordinated public health campaigns focused on risk reduction (like promoting safe recreational water use and proper chlorination) are vital for community-level prevention. By addressing these critical research and implementation gaps, the devastating outcomes associated with N. fowleri infections may be significantly mitigated, transforming a neglected and often fatal condition into a more preventable and treatable disease.
Acknowledgements
None
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
Abd El Wahab, W. M., El-Badry, A. A., & Hamdy, D. A. (2018). Molecular characterization and phylogenetic analysis of Acanthamoeba isolates in tap water of Beni-Suef, Egypt. Acta Parasitol, 63(4), 826-834. https://doi.org/10.1515/ap-2018-0101
Adler-Moore, J., Lewis, R. E., Brüggemann, R. J. M., Rijnders, B. J. A., Groll, A. H., & Walsh, T. J. (2019). Preclinical Safety, Tolerability, Pharmacokinetics, Pharmacodynamics, and Antifungal Activity of Liposomal Amphotericin B. Clinical Infectious Diseases, 68(Supplement_4), S244-S259. https://doi.org/10.1093/cid/ciz064
Ahmad, A. F., Lonnen, J., Andrew, P. W., & Kilvington, S. (2011). Development of a rapid DNA extraction method and one-step nested PCR for the detection of Naegleria fowleri from the environment. Water Res, 45(16), 5211-5217. https://doi.org/10.1016/j.watres.2011.07.025
AI-Herrawy, A., Mohamed, S., AI-Herrawy, A., Mohammed, A., & Gad, M. (2014). Distribution of Naegleria in water resources in Egypt Egyptian J Environ Res, 2, 1-14.
Alanazi, A., Younas, S., Ejaz, H., Alruwaili, M., Alruwaili, Y., Mazhari, B. B. Z., Atif, M., & Junaid, K. (2025). Advancing the understanding of Naegleria fowleri: Global epidemiology, phylogenetic analysis, and strategies to combat a deadly pathogen. Journal of Infection and Public Health, 18(4), 102690. https://doi.org/https://doi.org/10.1016/j.jiph.2025.102690
Ali, M., Jamal, S. B., & Farhat, S. M. (2020). Naegleria fowleri in Pakistan. Lancet Infect Dis, 20(1), 27-28. https://doi.org/10.1016/S1473-3099(19)30675-9
Anwar, A., Mungroo, M. R., Anwar, A., Sullivan, W. J., Jr., Khan, N. A., & Siddiqui, R. (2019). Repositioning of Guanabenz in Conjugation with Gold and Silver Nanoparticles against Pathogenic Amoebae Acanthamoeba castellanii and Naegleria fowleri. ACS Infectious Diseases, 5(12), 2039-2046. https://doi.org/10.1021/acsinfecdis.9b00263
Aurongzeb, M., Fatima, S. Z., Hussain, S. I., Rashid, Y., Aziz, T., Alhomrani, M., Alsanie, W. F., & Alamri, A. S. (2025). Detection and identification of Naegleria species along with Naegleria fowleri in the tap water samples. BMC Medical Genomics, 18(1), 6. https://doi.org/10.1186/s12920-024-02068-2
Aykur, M., & Dagci, H. (2021). Evaluation of molecular characterization and phylogeny for quantification of Acanthamoeba and Naegleria fowleri in various water sources, Turkey. PLoS One, 16(8), e0256659. https://doi.org/10.1371/journal.pone.0256659
Barnett, N. D., Kaplan, A. M., Hopkin, R. J., Saubolle, M. A., & Rudinsky, M. F. (1996). Primary amoebic meningoencephalitis with Naegleria fowleri: clinical review. Pediatr Neurol, 15(3), 230-234. https://doi.org/10.1016/s0887-8994(96)00173-7
Bartrand, T. A., Causey, J. J., & Clancy, J. L. (2014). Naegleria fowleri: An emerging drinking water pathogen. Journal AWWA, 106(10), E418-E432. https://doi.org/https://doi.org/10.5942/jawwa.2014.106.0140
Bellini, N. K., Santos, T. M., da Silva, M. T. A., & Thiemann, O. H. (2018). The therapeutic strategies against Naegleria fowleri. Exp Parasitol, 187, 1-11. https://doi.org/10.1016/j.exppara.2018.02.010
Bonilla-Lemus, P., Caballero Villegas, A. S., Carmona Jimenez, J., & Lugo Vazquez, A. (2014). Occurrence of free-living amoebae in streams of the Mexico Basin. Exp Parasitol, 145 Suppl, S28-33. https://doi.org/10.1016/j.exppara.2014.07.001
Capewell, L. G., Harris, A. M., Yoder, J. S., Cope, J. R., Eddy, B. A., Roy, S. L., Visvesvara, G. S., Fox, L. M., & Beach, M. J. (2015). Diagnosis, Clinical Course, and Treatment of Primary Amoebic Meningoencephalitis in the United States, 1937-2013. J Pediatric Infect Dis Soc, 4(4), e68-75. https://doi.org/10.1093/jpids/piu103
Cardenas-Zuniga, R., Silva-Olivares, A., Villalba-Magdaleno, J. A., Sanchez-Monroy, V., Serrano-Luna, J., & Shibayama, M. (2017). Amphotericin B induces apoptosis-like programmed cell death in Naegleria fowleri and Naegleria gruberi. Microbiology (Reading), 163(7), 940-949. https://doi.org/10.1099/mic.0.000500
CDC. (2024a, January 9, 2024). About Amebic Meningitis. Centers for Disease Control and Prevention (CDC). Retrieved July 31, 2025 from https://www.cdc.gov/meningitis/about/amebic-meningitis.html
CDC. (2024b, June 5, 2024). Free Living Amebic Infections. Centers for Disease Control and Prevention (CDC). Retrieved July 30, 2025 from https://www.cdc.gov/dpdx/freelivingamebic/
Christopher-Hennings, J., Erickson, G. A., Hesse, R. A., Nelson, E. A., Rossow, S., Scaria, J., & Slavic, D. (2019). Diagnostic Tests, Test Performance, and Considerations for Interpretation. In Diseases of Swine (pp. 75-97). https://doi.org/https://doi.org/10.1002/9781119350927.ch6
Cope, J. R., Conrad, D. A., Cohen, N., Cotilla, M., DaSilva, A., Jackson, J., & Visvesvara, G. S. (2016). Use of the Novel Therapeutic Agent Miltefosine for the Treatment of Primary Amebic Meningoencephalitis: Report of 1 Fatal and 1 Surviving Case. Clin Infect Dis, 62(6), 774-776. https://doi.org/10.1093/cid/civ1021
Datta, N. (2024). Molecular mechanisms and therapeutic strategies of Naegleria fowleri Carter (1970): a review of the fatal brain-eating amoeba. The Journal of V.N.Karazin Kharkiv National University. Series «Biology», 42, 22-31. https://doi.org/10.26565/2075-5457-2024-42-2
Debnath, A., Calvet, C. M., Jennings, G., Zhou, W., Aksenov, A., Luth, M. R., Abagyan, R., Nes, W. D., McKerrow, J. H., & Podust, L. M. (2017). CYP51 is an essential drug target for the treatment of primary amoebic meningoencephalitis (PAM). PLoS Negl Trop Dis, 11(12), e0006104.
Debnath, A., Tunac, J. B., Galindo-Gomez, S., Silva-Olivares, A., Shibayama, M., & McKerrow, J. H. (2012). Corifungin, a new drug lead against Naegleria, identified from a high-throughput screen. Antimicrob Agents Chemother, 56(11), 5450-5457. https://doi.org/10.1128/AAC.00643-12
Değerli, S., Değerli, N., Çamur, D., Doğan, Ö., & İlter, H. (2020). Genotyping by Sequencing of Acanthamoeba and Naegleria Isolates from the Thermal Pool Distributed Throughout Turkey. Acta Parasitologica, 65(1), 174-186. https://doi.org/10.2478/s11686-019-00148-3
Di Filippo, M. M., Novelletto, A., Di Cave, D., & Berrilli, F. (2017). Identification and phylogenetic position of Naegleria spp. from geothermal springs in Italy. Experimental Parasitology, 183, 143-149.
Di Terlizzi, R., & Platt, S. R. (2009). The function, composition and analysis of cerebrospinal fluid in companion animals: part II - analysis. Vet J, 180(1), 15-32. https://doi.org/10.1016/j.tvjl.2007.11.024
Diaz, J. (2012). Seasonal primary amebic meningoencephalitis (PAM) in the south: summertime is PAM time. The Journal of the Louisiana State Medical Society, 164(3), 148-155.
Dorlo, T. P., Balasegaram, M., Beijnen, J. H., & de Vries, P. J. (2012). Miltefosine: a review of its pharmacology and therapeutic efficacy in the treatment of leishmaniasis. J Antimicrob Chemother, 67(11), 2576-2597. https://doi.org/10.1093/jac/dks275
Elseadawy, R., Abbas, I., Al-Araby, M., & Abu-Elwafa, S. (2023). Occurrence and molecular characterization of Acanthamoeba, Naegleria fowleri and Blastocystis in water samples from various sources in Egypt. Acta Trop, 237, 106733. https://doi.org/10.1016/j.actatropica.2022.106733
Elsheikha, H. M., Siddiqui, R., & Khan, N. A. (2020). Drug Discovery against Acanthamoeba Infections: Present Knowledge and Unmet Needs. Pathogens, 9(5), 405. https://www.mdpi.com/2076-0817/9/5/405
Gabriel, S., Khan, N. A., & Siddiqui, R. (2019). Occurrence of free-living amoebae (Acanthamoeba, Balamuthia, Naegleria) in water samples in Peninsular Malaysia. J Water Health, 17(1), 160-171. https://doi.org/10.2166/wh.2018.164
Ghanchi, N. (2018). Increasing cases of Naegleria fowleri infections from Karachi Pakistan. Int J Infect Dis, 73, 185.
Gharpure, R., Bliton, J., Goodman, A., Ali, I. K. M., Yoder, J., & Cope, J. R. (2020). Epidemiology and Clinical Characteristics of Primary Amebic Meningoencephalitis Caused by Naegleria fowleri: A Global Review. Clinical Infectious Diseases, 73(1), e19-e27. https://doi.org/10.1093/cid/ciaa520
Goswick, S. M., & Brenner, G. M. (2003). Activities of azithromycin and amphotericin B against Naegleria fowleri in vitro and in a mouse model of primary amebic meningoencephalitis. Antimicrob Agents Chemother, 47(2), 524-528. https://doi.org/10.1128/AAC.47.2.524-528.2003
Grace, E., Asbill, S., & Virga, K. (2015). Naegleria fowleri: pathogenesis, diagnosis, and treatment options. Antimicrob Agents Chemother, 59(11), 6677-6681. https://doi.org/10.1128/AAC.01293-15
Gunarathna, N., Amarasinghe, A., Wijesundara, S., Iddawela, D., & Wickramasinghe, S. (2021). Isolation, molecular characterization and phylogeny of Naegleria species in water bodies of North-Western Province, Sri Lanka. PLoS One, 16(3), e0248510. https://doi.org/10.1371/journal.pone.0248510
Hannisch, W., & Hallagan, L. F. (1997). Primary amebic meningoencephalitis: a review of the clinical literature. Wilderness Environ Med, 8(4), 211-213. https://doi.org/10.1580/1080-6032(1997)008[0211:pamaro]2.3.co;2
Heggie, T. W., & Kupper, T. (2017). Surviving Naegleria fowleri infections: A successful case report and novel therapeutic approach. Travel Med Infect Dis, 16, 49-51. https://doi.org/10.1016/j.tmaid.2016.12.005
Huang, S. W., & Hsu, B. M. (2010). Survey of Naegleria and its resisting bacteria-Legionella in hot spring water of Taiwan using molecular method. Parasitol Res, 106(6), 1395-1402. https://doi.org/10.1007/s00436-010-1815-0
Hundt, M. J., & Ruffolo, C. G. (2005). Interaction of Pasteurella multocida with free-living amoebae. Appl Environ Microbiol, 71(9), 5458-5464. https://doi.org/10.1128/AEM.71.9.5458-5464.2005
Khalifa, R. M., Ahmad, A. K., Abdel-Hafeez, E. H., & Mosllem, F. A. (2014). Present status of protozoan pathogens causing water-borne disease in northern part of El-Minia Governorate, Egypt. J Egypt Soc Parasitol, 44(3), 559-566. https://doi.org/10.12816/0007860
Kidney, D. D., & Kim, S. H. (1998). CNS infections with free-living amebas: neuroimaging findings. AJR Am J Roentgenol, 171(3), 809-812. https://doi.org/10.2214/ajr.171.3.9725321
Kim, J.-H., Jung, S.-Y., Lee, Y.-J., Song, K.-J., Kwon, D., Kim, K., Park, S., Im, K.-I., & Shin, H.-J. (2008). Effect of therapeutic chemical agents in vitro and on experimental meningoencephalitis due to Naegleria fowleri. Antimicrob Agents Chemother, 52(11), 4010-4016.
Kim, J.-H., Lee, Y.-J., Sohn, H.-J., Song, K.-J., Kwon, D., Kwon, M.-H., Im, K.-I., & Shin, H.-J. (2008). Therapeutic effect of rokitamycin in vitro and on experimental meningoencephalitis due to Naegleria fowleri. Int J Antimicrob Agents, 32(5), 411-417.
Kim, M.-j., Lee, G.-C., Kim, K., Lee, H., Kim, M. Y., Seo, D. K., Lee, J. Y., & Cho, Y.-C. (2018). Surveillance of Acanthamoeba spp. and Naegleria fowleri in environmental water by using the duplex real-time PCR. Microbiolog Soc Korea, 54(2), 98-104.
Krishnamoorthi, S., Sharma, C., Mewara, A., & Khurana, S. (2022). Environmental water surveillance for free-living amoeba in North India. Indian J Med Microbiol, 40(3), 389-393. https://doi.org/10.1016/j.ijmmb.2022.05.002
Kurup, S. B., Lekshmi, P., & Vijayan, S. (2022). Naegleria fowleri-the brain-eating amoeba: A review. World J Pharm Res, 11(1), 426-440.
Laniado-Laborin, R., & Cabrales-Vargas, M. N. (2009). Amphotericin B: side effects and toxicity. Rev Iberoam Micol, 26(4), 223-227. https://doi.org/10.1016/j.riam.2009.06.003
Laseke, I., Korte, J., Lamendella, R., Kaneshiro, E. S., Marciano-Cabral, F., & Oerther, D. B. (2010). Identification of Naegleria fowleri in warm ground water aquifers. J Environ Qual, 39(1), 147-153. https://doi.org/10.2134/jeq2009.0062
Latifi, A. R., Niyyati, M., Lorenzo-Morales, J., Haghighi, A., Tabaei, S. J., Lasjerdi, Z., & Azargashb, E. (2017). Occurrence of Naegleria species in therapeutic geothermal water sources, Northern Iran. Acta Parasitol, 62(1), 104-109. https://doi.org/10.1515/ap-2017-0012
Maciver, S. K., Piñero, J. E., & Lorenzo-Morales, J. (2020). Is Naegleria fowleri an emerging parasite? Trends in parasitology, 36(1), 19-28.
Maclean, R. C., Richardson, D. J., LePardo, R., & Marciano-Cabral, F. (2004). The identification of Naegleria fowleri from water and soil samples by nested PCR. Parasitol Res, 93(3), 211-217. https://doi.org/10.1007/s00436-004-1104-x
Madarova, L., Trnkova, K., Feikova, S., Klement, C., & Obernauerova, M. (2010). A real-time PCR diagnostic method for detection of Naegleria fowleri. Exp Parasitol, 126(1), 37-41. https://doi.org/10.1016/j.exppara.2009.11.001
Mahajan, A., & Mahajan, A. (2024). Neuroimaging: CT Scan and MRI. In H. Prabhakar, V. Singhal, K. G. Zirpe, & H. Sapra (Eds.), Principles and Practice of Neurocritical Care (pp. 189-215). Springer Nature Singapore. https://doi.org/10.1007/978-981-99-8059-8_14
Marciano-Cabral, F., & Cabral, G. A. (2007). The immune response to Naegleria fowleri amebae and pathogenesis of infection. FEMS Immunol Med Microbiol, 51(2), 243-259. https://doi.org/10.1111/j.1574-695X.2007.00332.x
Marciano-Cabral, F., Cline, M. L., & Bradley, S. G. (1987). Specificity of antibodies from human sera for Naegleria species. J Clin Microbiol, 25(4), 692-697. https://doi.org/10.1128/jcm.25.4.692-697.1987
Martinez, A. (1985). Free-living amebas: natural history, pre-vention, diagnosis, pathology and treatment of disease. CRC Press.
Martinez, A. J. (2019). Free-Living Amebas: Natural History, Prevention, Diagnosis, Pathology, and Treatment of Disease. CRC Press.
Martínez, A. J., García, C. A., Halks-Miller, M., & Arce-Vela, R. (1980). Granulomatous amebic encephalitis presenting as a cerebral mass lesion. Acta neuropatholog, 51, 85-91.
Martinez, A. J., & Visvesvara, G. S. (1991). Laboratory diagnosis of pathogenic free-living amoebas: Naegleria, Acanthamoeba, and Leptomyxid. Clin Lab Med, 11(4), 861-872. https://www.ncbi.nlm.nih.gov/pubmed/1802525
Martinez, A. J., & Visvesvara, G. S. (1997). Free-living, amphizoic and opportunistic amebas. Brain Pathol, 7(1), 583-598. https://doi.org/10.1111/j.1750-3639.1997.tb01076.x
Milanez, G., Masangkay, F., Somsak, V., Kotepui, M., Tangpong, J., & Karanis, P. (2019). Occurrence and the first report of Naegleria australiensis presence in a major lake in the Philippines. J Water Health, 17(4), 647-653. https://doi.org/10.2166/wh.2019.034
Muchesa, P., Leifels, M., Jurzik, L., Hoorzook, K. B., Barnard, T. G., & Bartie, C. (2017). Coexistence of free-living amoebae and bacteria in selected South African hospital water distribution systems. Parasitol Res, 116(1), 155-165. https://doi.org/10.1007/s00436-016-5271-3
Mungroo, M. R., Anwar, A., Khan, N. A., & Siddiqui, R. (2020). Gold-Conjugated Curcumin as a Novel Therapeutic Agent against Brain-Eating Amoebae. ACS Omega, 5(21), 12467-12475. https://doi.org/10.1021/acsomega.0c01305
Mungroo, M. R., Khan, N. A., & Siddiqui, R. (2019). Naegleria fowleri: diagnosis, treatment options and pathogenesis. Expert Opinion on Orphan Drugs, 7(2), 67-80. https://doi.org/10.1080/21678707.2019.1571904
Naqvi, A. A., Yazdani, N., Ahmad, R., Zehra, F., & Ahmad, N. (2016). Epidemiology of primary amoebic meningoencephalitis-related deaths due to Naegleria fowleri infections from freshwater in Pakistan: An analysis of 8-year dataset. Arch Pharm Practice, 7(4).
Painter, S. M., Pfau, R. S., Brady, J. A., & McFarland, A. M. (2013). Quantitative assessment of Naegleria fowleri and Escherichia coli concentrations within a Texas reservoir. J Water Health, 11(2), 346-357.
Phung, N. T. N., Pham, H. T., Tran, T. T., Dinh, V. H., Tran, N. M., Tran, N. A. N., Ngo, M. Q. N., Nguyen, H. T. T., Tran, D. K., Le, T. K. T., Quek, C., Pham, V. H., & Pham, S. T. (2025). Naegleria fowleri: Portrait of a Cerebral Killer. Diagnostics, 15(1), 89. https://www.mdpi.com/2075-4418/15/1/89
Reveiller, F. L., Varenne, M. P., Pougnard, C., Cabanes, P. A., Pringuez, E., Pourima, B., Legastelois, S., & Pernin, P. (2003). An enzyme-linked immunosorbent assay (ELISA) for the identification of Naegleria fowleri in environmental water samples. J Eukaryot Microbiol, 50(2), 109-113. https://doi.org/10.1111/j.1550-7408.2003.tb00244.x
Reyes-Batlle, M., Rizo-Liendo, A., Viera-Santana, R. A., Afonso-Morales, S., Lopez-Arencibia, A., Sifaoui, I., Chiboub, O., Bethencourt-Estrella, C. J., Nicolas-Hernandez, D. S., Rodriguez-Exposito, R. L., Zamora-Herrera, J., Valladares, B., Pinero, J. E., Diaz, F. J., & Lorenzo-Morales, J. (2019). Isolation and Molecular Identification of Naegleria australiensis in Irrigation Water of Fuerteventura Island, Spain. Acta Parasitol, 64(2), 331-335. https://doi.org/10.2478/s11686-019-00046-8
Reyes-Batlle, M., Wagner, C., Lopez-Arencibia, A., Sifaoui, I., Martinez-Carretero, E., Valladares, B., Pinero, J. E., & Lorenzo-Morales, J. (2017). Isolation and molecular characterization of a Naegleria strain from a recreational water fountain in Tenerife, Canary Islands, Spain. Acta Parasitol, 62(2), 265-268. https://doi.org/10.1515/ap-2017-0033
Rice, C. A., Colon, B. L., Chen, E., Hull, M. V., & Kyle, D. E. (2020). Discovery of repurposing drug candidates for the treatment of diseases caused by pathogenic free-living amoebae. PLoS Negl Trop Dis, 14(9), e0008353. https://doi.org/10.1371/journal.pntd.0008353
Saberi, R., Seifi, Z., Dodangeh, S., Najafi, A., Abdollah Hosseini, S., Anvari, D., Taghipour, A., Norouzi, M., & Niyyati, M. (2020). A systematic literature review and meta-analysis on the global prevalence of Naegleria spp. in water sources. Transbound Emerg Dis, 67(6), 2389-2402. https://doi.org/10.1111/tbed.13635
Schumacher, D. J., Tien, R. D., & Lane, K. (1995). Neuroimaging findings in rare amebic infections of the central nervous system. AJNR Am J Neuroradiol, 16(4 Suppl), 930-935. https://www.ncbi.nlm.nih.gov/pubmed/7611077
Schuster, F. L. (2002). Cultivation of pathogenic and opportunistic free-living amebas. Clin Microbiol Rev, 15(3), 342-354. https://doi.org/10.1128/CMR.15.3.342-354.2002
Seehusen, D. A., Reeves, M. M., & Fomin, D. A. (2003). Cerebrospinal fluid analysis. Am Fam Physician, 68(6), 1103-1108. https://www.ncbi.nlm.nih.gov/pubmed/14524396
Shirsath, S. S., Shirsath, V. S., & Bhopale, R. T. (2024). Naegleria fowleri 98 Percent Fatal a Comprehensive Survey. Research Journal of Science and Technology, 16(2), 163-168.
Siddiqui, R., & Khan, N. A. (2014). Primary amoebic meningoencephalitis caused by Naegleria fowleri: an old enemy presenting new challenges. PLoS Negl Trop Dis, 8(8), e3017. https://doi.org/10.1371/journal.pntd.0003017
Siddiqui, R., Lloyd, D., Alharbi, A. M., & Khan, N. A. (2024). Emerging therapies against Naegleria fowleri. Expert Opinion on Orphan Drugs, 12(1), 41-49. https://doi.org/10.1080/21678707.2024.2383173
Siddiqui, R., Maciver, S. K., & Khan, N. A. (2025). Naegleria fowleri: emerging therapies and translational challenges. Expert Review of Anti-infective Therapy, 1-9. https://doi.org/10.1080/14787210.2025.2536827
Singh, P., Kochhar, R., Vashishta, R. K., Khandelwal, N., Prabhakar, S., Mohindra, S., & Singhi, P. (2006). Amebic meningoencephalitis: spectrum of imaging findings. AJNR Am J Neuroradiol, 27(6), 1217-1221. https://www.ncbi.nlm.nih.gov/pubmed/16775267
Sriram, N., & Chandra, A. S. (2024). Brain-Eating Amoeba: A Comprehensive Review of Naegleria fowleri Infections and Primary Amoebic Meningoencephalitis. Scientific Hub of Applied Research in Emerging Medical science & technology, 3(5), 1-6.
Stahl, L. M., & Olson, J. B. (2020). Environmental abiotic and biotic factors affecting the distribution and abundance of Naegleria fowleri. FEMS Microbiology Ecology, 97(1). https://doi.org/10.1093/femsec/fiaa238
Stahl, L. M., Snead, A. A., & Olson, J. B. (2025). Predicting Naegleria fowleri freshwater habitat suitability in the United States for the present-day and future using ecological niche modeling. Water Research, 285, 124018. https://doi.org/https://doi.org/10.1016/j.watres.2025.124018
Tabassum, S., Naeem, A., Gill, S., Mumtaz, N., Khan, M. Z., Tabassum, S., Naeem, R., & Mukherjee, D. (2022). Increasing cases of Naegleria fowleri during the time of COVID 19; an emerging concern of Pakistan. Int J Surg, 105, 106881. https://doi.org/10.1016/j.ijsu.2022.106881
Tanveer, T., Hameed, A., Bin-Dukhyil, A.-A. A., Alaidarous, M., & Matin, A. (2017). Isolation of thermotolerant pathogenic Naegleria australiensis from diverse water resources including household drinking water in Pakistan. Acta Protozoologica, 56(4), 317-322.
Tiewcharoen, S., Junnu, V., Roongruangchai, K., Angkanasinsiri, A., & Rabablert, J. (2018). Molecular Identification of Naegleria fowleri and Pathogenic Acanthamoeba spp. in Chao Phraya River and Canals Around Siriraj Hospital, Thailand. Journal of the Medical Association of Thailand, 101(10).
Tiewcharoen, S., Komalamisra, N., & Junnu, V. (2004). Zymogram patterns of Naegleria spp isolated from natural water sources in Taling Chan district, Bangkok. Southeast Asian J Trop Med Public Health, 35(2), 275-280. https://www.ncbi.nlm.nih.gov/pubmed/15691124
Torrado, J. J., Espada, R., Ballesteros, M. P., & Torrado-Santiago, S. (2008). Amphotericin B formulations and drug targeting. J Pharm Sci, 97(7), 2405-2425. https://doi.org/10.1002/jps.21179
Trabelsi, H., Dendana, F., Sellami, A., Sellami, H., Cheikhrouhou, F., Neji, S., Makni, F., & Ayadi, A. (2012). Pathogenic free-living amoebae: epidemiology and clinical review. Pathol Biol (Paris), 60(6), 399-405. https://doi.org/10.1016/j.patbio.2012.03.002
Vargas-Zepeda, J., Gomez-Alcala, A. V., Vasquez-Morales, J. A., Licea-Amaya, L., De Jonckheere, J. F., & Lares-Villa, F. (2005). Successful treatment of Naegleria fowleri meningoencephalitis by using intravenous amphotericin B, fluconazole and rifampicin. Arch Med Res, 36(1), 83-86. https://doi.org/10.1016/j.arcmed.2004.11.003
Verweij, J., Planting, A., van der Burg, M., & Stoter, G. (1992). A dose-finding study of miltefosine (hexadecylphosphocholine) in patients with metastatic solid tumours. J Cancer Res Clin Oncol, 118(8), 606-608. https://doi.org/10.1007/BF01211805
Visvesvara, G. S. (2013). Infections with free-living amebae. Handb Clin Neurol, 114, 153-168. https://doi.org/10.1016/B978-0-444-53490-3.00010-8